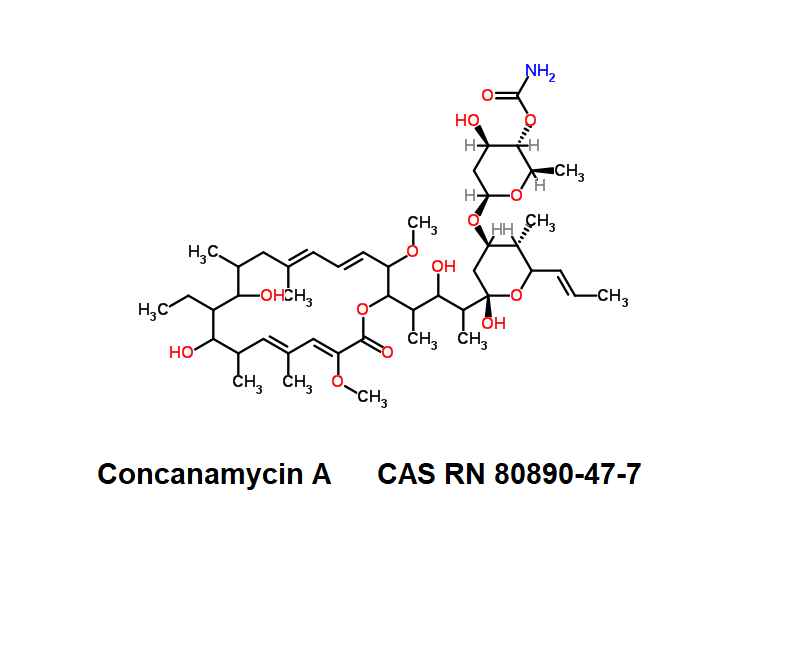
Concanamycin A (Folimycin)
Details
Specifications
Chemical identification
Synonyms:
- Concamycin A
- Folimycin
- Concanamycin
Comment: Many other suppliers, as well as PUBCHEM, CHEMSPIDER and publications, consider Folimycin and Concanamycin A to be synonyms, while SCIFINDER considers these compounds to be different.
IUPAC:
3-O-(4-O-Carbamoyl-2,6-dideoxy-β-D-arabino-hexopyranosyl)-2,4-dideoxy-1-C-{4-[(4E,6E,14E,16Z)-11-ethyl-10,12-dihydroxy-3,17-dimethoxy-7,9,13,15-tetramethyl-18-oxooxacyclooctadeca-4,6,14,16-tetraen- 2-yl]-3-hydroxy-2-pentanyl}-4-methyl-5-[(1E)-1-propen-1-yl]-β-D-threo-pentopyranose
RTECS# CB9732000
Macrolide type antibiotic, ATPase inhibitor, having antiproliferative, antifungal and antimalarial effects.
Further Information
Concanamycin A is reported to be soluble in chloroform, methanol, ethanol, acetone, ethyl acetate, and DMSO. Concanamycin A is not soluble in water.
NotGMO
Product natural
Composition
Special Info
Other Fields