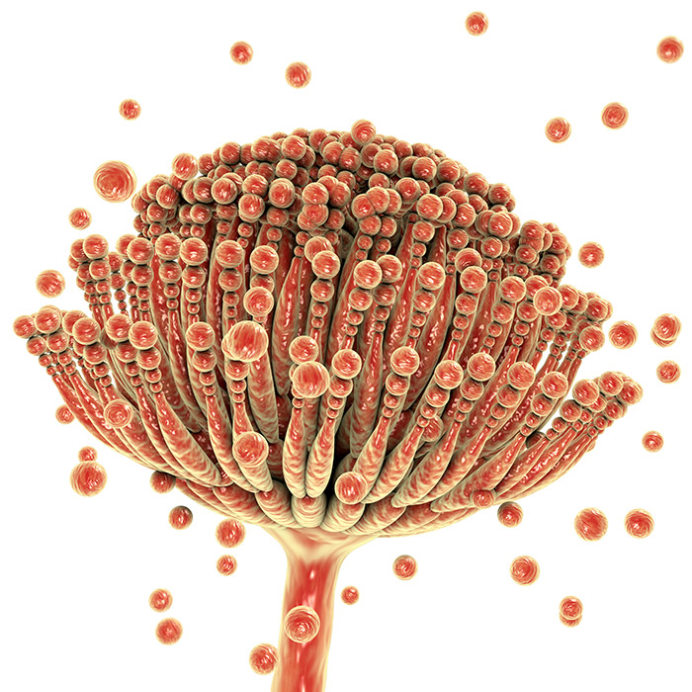
Mycotoxins are defined as naturally occurring substances produced by filamentous fungi (moulds) that arouse toxic response when introduced in low amounts to animals through a natural pathway. These fungi are referred to as “toxigenic” fungi. They grow naturally as a form of contaminants on some food crops, majorly cereals, nuts and fruit. Conventionally, toxigenic fungi which contaminate crops have been classified into two groups: “field” and “storage” fungi. Field fungi (such as Cladosporium, Fusarium and Alternaria spp.) get to plants during the developmental stage of the plant; while storage fungi (e.g., Aspergillus and Penicillium spp.) proliferate during the storage period.
Mycotoxins, which are low weight secondary metabolites, produce harmful substances called Mycotoxicoses in animals which invariably impact public health. The toxicity effects of Mycotoxins could be acute or chronic, or both. The most toxic of these, is Aflatoxins. Aflatoxins B1, B2, G1 and G2 are a critical concern. A low-level exposure to Aflatoxin for a long term has been associated with different liver diseases such as cancer, jaundice, cirrhosis and hepatis. Also, they are genotoxic (DNA-damaging) carcinogenic and immunosuppressants. Ochratoxin A is another type of mycotoxin that is teratogenic (reproductive), immunosuppressant, and had been proved to have connection with kidney diseases. Other toxigenic fungi are associated with protein synthesis inhibition, genotoxic, and immune dysfunction.
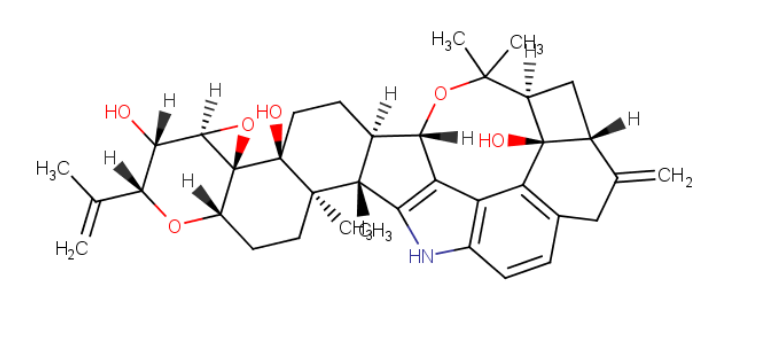
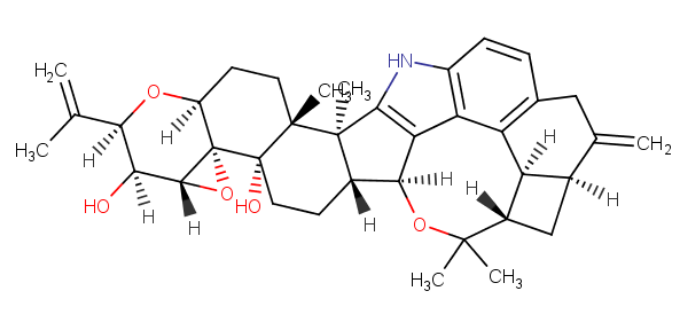
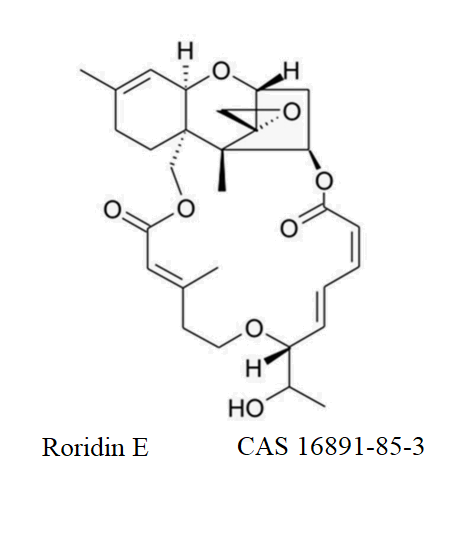
Fermentek is a global leader in the production of high-standard reference material for Mycotoxins for food safety analysis and research. The company offers more than 60 types of Mycotoxins manufactured from various different microorganisms, divided into nine known Mycotoxin families. These products are formulated into a powdered form or standard solutions with high purity characterized by their melting points, TLC & HPLC.