Melittin: a major peptide component of bee venom, its various effects and the mechanisms of its actions.
Melittin, is the main constituent in the venom of the European honeybee Apis mellifera. It has attracted considerable attention among researchers, owing to its potential therapeutic applications1.
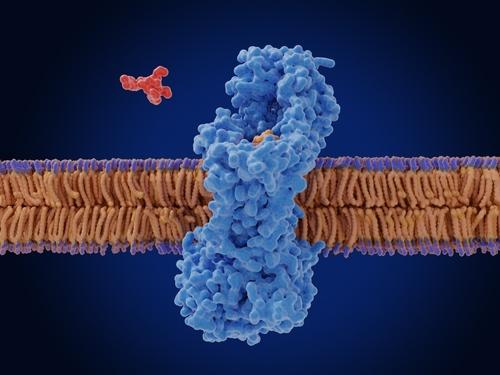
Melittin is a cationic water-soluble linear peptide-amide, containing 26 amino acid residues2. It acts as an archetypal membrane-active peptide, and is hydrophobic and amphipathic, which drives strong partitioning into membranes2. Membrane binding is coupled to folding into α-helical secondary structure, which results in the insertion of amphipathic Melittin into a bilayer, thus dramatically reducing the permeability barrier by disrupting the strict segregation of polar, and non-polar moieties across the bilayer. In solution, Melittin molecules are monomeric and have random coil structure at low concentration2.
At high concentration, Melittin folds into α-helical tetramers in which the hydrophobic surfaces of the amphipathic helices are buried, and the polar surfaces are exposed2. When Melittin permeabilizes a biological membrane, water, ions, metabolites and other molecules pass freely through the membrane and the cell is rapidly killed2.
Melittin is a toxic peptide. Under certain conditions, Melittin at low concentrations, when conjugated to proteins or to other molecules, or formulated in nanoparticles or liposomes, reduces its undesirable effects. Its activity has been characterized in a broad array of systems, both in vitro and in vivo. It has been tested against host and pathogen cells from all branches of life, against infectious diseases and disease pathogens, and against cancer, and many other diseases from many different tissues2. An extremely wide array of interesting and potentially useful biological activities has been reported for Melittin:
- Anti-bacterial activity - The anti-bacterial activity of bee venom was first reported in 19412. Melittin has anti-bacterial effects against penicillin-resistant Staphylococcus aureus2. Melittin was also shown to have activity against clinical isolates of gram-negative as well as gram-positive bacteria2.
- Anti-viral activity - Melittin has potent broad-spectrum inhibitory activity against both enveloped and nonenveloped viruses2.
- Anti-fungal activity - Melittin has inhibitory and antifungal effects at micromolar concentrations against a broad spectrum of fungal genera, including Candida albicans, one of the main pathogens responsible for human fungal infections2. Melittin hinders fungal growth by several mechanisms such as membrane permeabilization, apoptosis induction by reactive oxygen species-mediated mitochondria/caspase-dependent pathway, inhibition of (1,3)-β-D-glucan synthase, and alterations in fungal gene expression1.
- Anti-Parasital activity - Melittin has direct antiparasitic effects against the sporogonic and gametocyte stages of the Plasmodium life cycle. Melittin has also been tested against Leishmania parasites, the causative agent of leishmaniasis. Melittin demonstrated antiparasitic activity against Toxoplasma gondi2.
- Anti-Eukaryotic cell activity - Melittin acts as a non-specific lytic peptide with a broad-spectrum activity against eukaryotic cells2. The lytic effect of Melittin has been shown to arise from its ability to disrupt plasma membrane by insertion into phospholipid bilayers. Melittin interacts with mammalian cells to cause plasma membrane disruption. For example, in vitro treatment of gastrointestinal epithelial cell lines with Melittin results in loss of plasma membrane integrity even in low concentrations. The peptide is highly toxic to the epithelial cells, endothelial cells, inhibits the proliferation of vascular smooth muscle cells, exerts cytotoxic effects and inhibits the proliferation of hepatic cells by inducing apoptotic cell death2.
- Anti-Cancer activity - In the cell and animal models and in vitro experiments, Melittin has been shown to affect cell proliferation, apoptosis, and cancer cell metastasis, all of which indicate a possible use of Melittin or Melittin-modified peptides for cancer treatment. A variety of cancer cells were found sensitive to low concentration range of Melittin (e.g. lung, breast, ovarian, osteosarcoma cells, gastric adenocarcinoma cells, promyelocytic leukemic granulocytes, breast cancer cell motility and migration, melanoma cells, etc)3. Numerous research groups have adopted different approaches, including conjugation with tumor-targeting proteins, gene therapy, and encapsulation of Melittin in nanoparticles to reduce the non-specific cytotoxic effects of Melittin and potentiate its anti-cancerous activity, as well as creating various hybrid peptides based on Melittin3,4,5.
Other activities - Paracellular tight junctions Blood brain barrier (BBB) opening, immune modulation, analgesic and anti-inflammatory effects, dermatological effects (positive effects on Atopic dermatitis in vivo) and more2.
Beside activities on eukaryotic cells, Melittin was also found to affect invertebrate animals, as well as plants tissues and to increase plant protection from pathogens2.
Due to its vast activities, the interest in Melittin is growing fast towards its future optimization and its variants in different formulations. Melittin forms transient pores which enable leakage from membranes only for a limited time after the peptide is added to bilayers2. Two generation of Melittin synthetic variant libraries were developed and tested to achieve the optimized Melittin with less toxicity and better activity. The first generation focuses on elucidating the best variants forming explicit pores in membranes2 and the second generation focuses on achieving a peptide that can be triggered to form macromolecule-sized pores only in tumors that could induce the selective apoptosis of malignant cells. The optimized peptide should be active at acidic pH, which is a marker of the tumor microenvironment and is also found in endosomes and lysosomes of endocytotic pathways2.
Fermentek offers high purity Melittin (MEL-001) in various sizes for research and analysis purposes. Our Melittin is a natural peptide, derived from the honeybee, Apis Mellifera and is available in various sizes, ready for immediate shipment.
1. Memariani H, Memariani M. Anti-Fungal Properties and Mechanisms Of Melittin. Appl Microbiol Biotechnol. 2020 Aug;104(15):6513-6526. doi: 10.1007/s00253-020-10701-0. Epub 2020 Jun 4. PMID: 32500268.
2. Guha S, Ferrie RP, Ghimire J, Ventura CR, Wu E, Sun L, Kim SY, Wiedman GR, Hristova K, Wimley WC. Applications and Evolution of Melittin, the Quintessential Membrane Active Peptide. Biochem Pharmacol. 2021 Nov;193:114769. doi: 10.1016/j.bcp.2021.114769. Epub 2021 Sep 17. PMID: 34543656; PMCID: PMC9235364.
3. Haque S, Hussain A, Joshi H, Sharma U, Sharma B, Aggarwal D, Rani I, Ramniwas S, Gupta M, Tuli HS. Melittin: a Possible Regulator of Cancer Proliferation in Preclinical Cell Culture and Animal Models. J Cancer Res Clin Oncol. 2023 Dec;149(19):17709-17726. doi: 10.1007/s00432-023-05458-8. Epub 2023 Nov 3. PMID: 37919474.
4. Wang A, Zheng Y, Zhu W, Yang L, Yang Y, Peng J. Melittin-Based Nano-Delivery Systems for Cancer Therapy. Biomolecules. 2022 Jan 12;12(1):118. doi: 10.3390/biom12010118. PMID: 35053266; PMCID: PMC8773652.
5. Duffy, C., Sorolla, A., Wang, E. et al. Honeybee Venom and Melittin Suppress Growth Factor Receptor Activation in HER2-Enriched and Triple-Negative Breast Cancer. npj Precis. Onc. 4, 24 (2020).https://doi.org/10.1038/s41698-020-00129-0